GMP Peptide Manufacturing Overview
With 25 years of dedicated service to the pharmaceutical industry, CPC Scientific specializes in large-scale, late-phase, and commercial GMP peptide manufacturing. As a leading CDMO with an annual peptide API production capacity of over 1,000 kg and a per-batch peptide production capacity of 20–25 kg, we manufacture synthetic GMP peptides across early-phase development, late-phase development, and full commercial production, providing complete lifecycle support.
Our production facilities adhere rigorously to CGMP guidelines as mandated by major global regulatory authorities, including the NMPA, FDA, EMA, TGA, and MFDS. We have successfully passed FDA inspections five times as of the Latest Practicable Date. Furthermore, our facility holds ISO9001 and ISO13485 certifications for quality management systems.
Late Phase GMP Production Track Record
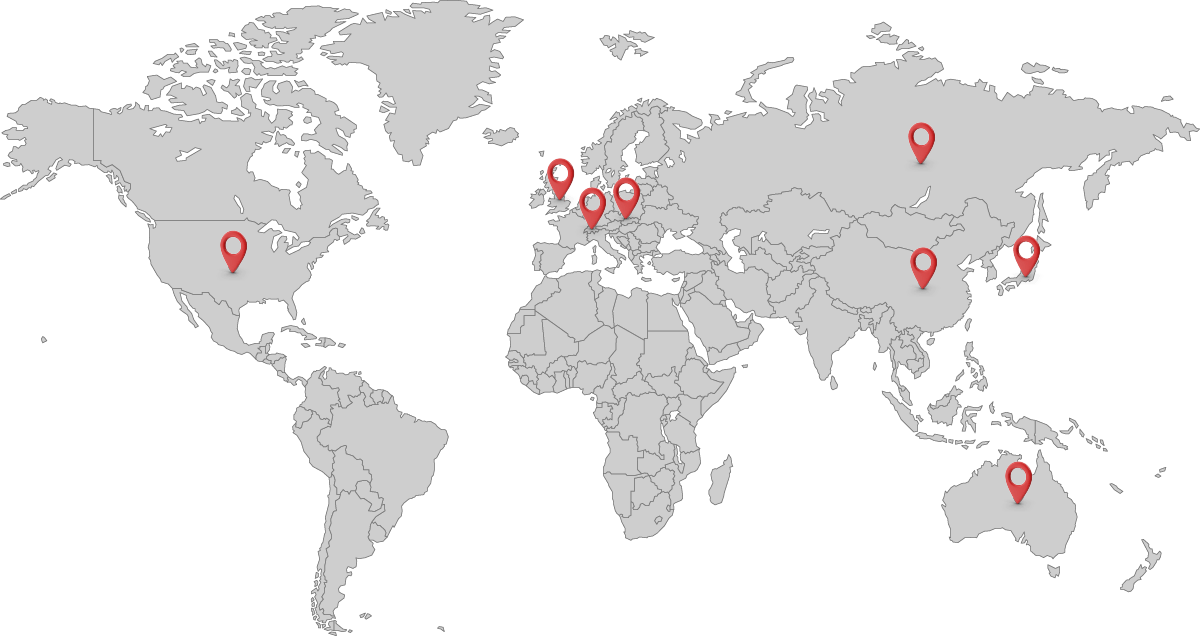
CGMP Peptide Manufacturing Projects by Region
| Project Location | Development Stage | |
|---|---|---|
| USA | Approved | |
| Japan | Approved | |
| Europe Union | Approved | |
| UK | Approved | |
| Switzerland | Approved | |
| China | Approved | |
| Australia | Approved | |
| Russia and CIS | Approved | |
Our production facilities adhere rigorously to CGMP guidelines as mandated by major global regulatory authorities, including the NMPA, FDA, EMA, TGA, and MFDS. We have successfully passed FDA inspections five times as of the Latest Practicable Date.
Furthermore, our facility holds ISO9001 and ISO13485 certifications for quality management systems.
WHY CHOOSE CPC SCIENTIFIC?
We understand that there are many peptide CDMO choices in today’s market. Learn more about what separates CPC Scientific from other GMP manufacturers.
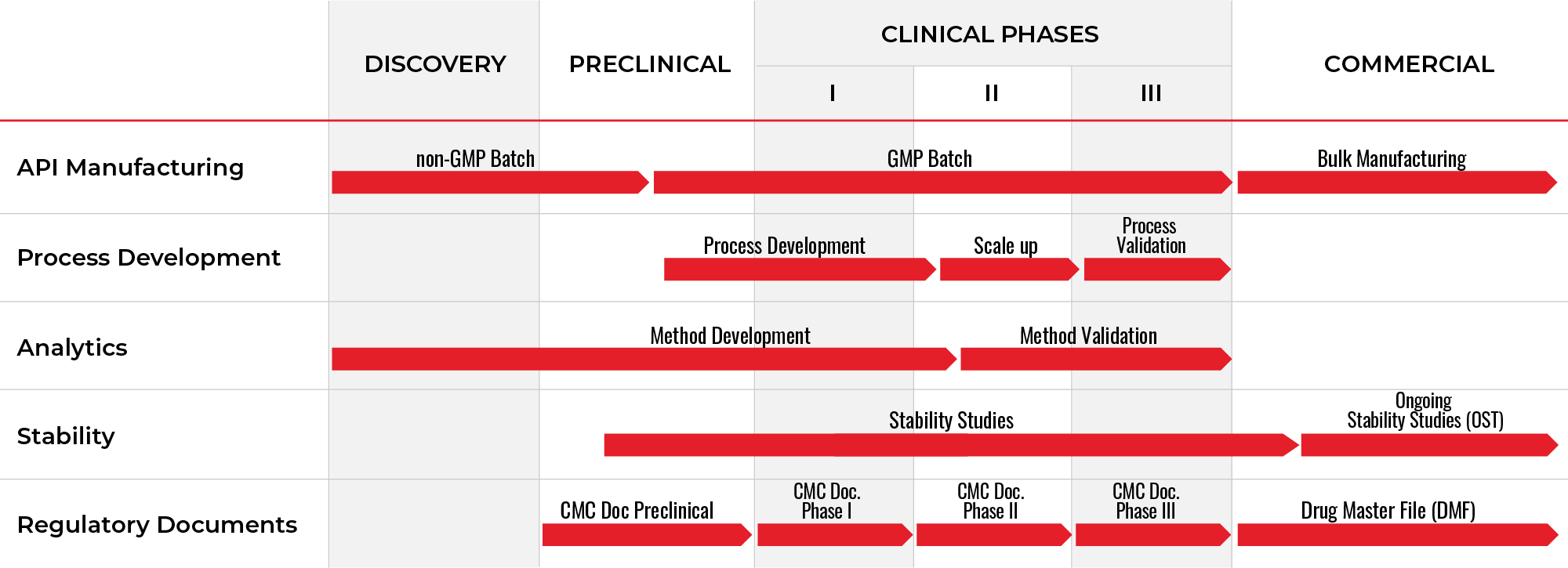
COMPLETE LIFECYCLE SUPPORT FOR GMP PEPTIDES
CPC Scientific’s integrated service model is designed to support partners through the most critical phases of GMP peptide drug development, with particular focus on late-phase development, large-scale manufacturing, and commercial production.
GMP Peptide Manufacturing Brochure Request
With 25 years of committed service to the peptide industry, CPC Scientific is a trusted peptide manufacturer of high-quality APIs to pharmaceutical companies and researchers all over the world. We are comprised of peptide experts who have actively pursued peptide design and large scale manufacturing for more than 30 years. Discover how we can support your GMP peptide and oligonucleotide manufacturing needs throughout the therapeutic lifecycle with our comprehensive CRDMO services.

Explore Our Expertise in GMP Peptides
Latest News on GMP Peptides
CPC Scientific’s recent articles and resources on GMP peptides
CPC Scientific today announced that its Hangzhou-based CGMP manufacturing facility has successfully completed the DMF filing for its self-developed Tirzepatide API with the U.S. Food and Drug Administration...
Dive into Joseph Denby's presentation at NextGen Biomed 2026, where he shares a case study on delivering sustainable, efficient peptide manufacturing at scale through our advanced SPPS process optimizations.
CPC Scientific had a fantastic time connecting with partners, clients, and colleagues this year at DCAT Week 2026. See our highlights from New York, featuring generic API scale-up, the 98th DCAT Annual Dinner, and more...
Catch CPC Scientific's key moments from NextGen Biomed 2026, where we showcased how our advanced process design is accelerating production timelines and cost efficiency for greener TIDES manufacturing at scale...
Discover how CPC Scientific has earned the trust of top pharma and biotech companies across 50+ countries. See the numbers behind our GMP regulatory record, global project portfolio, clinical and commercial delivery timelines, and more.
CPC Scientific is thrilled to return to CPHI Frankfurt 2025! Explore our booth as we recap key moments from Day 1 of CPHI, where we're highlighting our position as a globally pioneering peptide & oligo CDMO...
GMP Peptide Manufacturing Citations
CPC Scientific peptide products manufactured under CGMP conditions
Nakagawa, Mayumi, Teresa Evans, Milan Bimali, Hannah Coleman, Jasmine Crane, Nadia Darwish, Jennifer L. Faulkner et al. MedRxiv (2025): 2025-01.
The vaccine consisted of four current good manufacturing production-grade synthetic peptides covering the HPV 16 E6 protein [amino acid (aa)1-45, 46-80, 81-115, and 116-158] (CPC Scientific, San Jose, CA [..]
Järveläinen, Harri A., Christian Schmithals, Maike von Harten, Bianca Kakoschky, Thomas J. Vogl, Stephen Harris, Claire Henson, Gemma Bullen-Clerkson, and Albrecht Piiper. International Journal of Molecular Sciences 24, no. 6 (2023): 5700.
The cyclic iRGD/CEND-1 peptide [sequence: CRGDKGPDC] and RGD control peptide (CRGDDGPKC) for pre-clinical use was sourced either from GenScript (Piscataway, NJ, USA) or CPC Scientific (Hangzhou, China).
Holt, Daniel P., Dhiraj Kumar, Sridhar Nimmagadda, and Robert F. Dannals. Journal of Labelled Compounds and Radiopharmaceuticals 66, no. 2 (2023): 47-54.
to current Good Manufacturing Practice (cGMP) requirements. In addition, the production is … -NODA; Figure 1) was custom synthesized from CPC Scientific (San Jose, CA). The authentic …
Axiak-Bechtel, Sandra M., Stacey B. Leach, David G. Scholten, Jessica R. Newton-Northup, Brendan J. Johnson, H. E. Durham, Kenneth A. Gruber, and Michael F. Callahan. Pharmacology Research & Perspectives 9, no. 3 (2021): e00777.
TCMCB07 [a cyclic substituted melanocortin antagonist with the structure Ac- Nle- cyclo[Asp- Pro- DNal(2’)-Arg- Trp- Lys]- DVal- DPro- NH2] was manufactured by CPC Scientific Inc. under cGMP conditions. Active pharmaceutical ingredient was dissolved in milliQ water at 10 mg ml−1, sterile filtered [..]”
SAN JOSE, CA., June 25, 2020 /CPCNewswire/ — CPC Scientific Inc., a San Jose based CDMO with GMP manufacturing facilities in Hangzhou, is pleased to announce that their drug master file (DMF) for cetrorelix acetate has been submitted to the U.S. Food and Drug Administration and has been issued DMF# 034884. This generic peptide API […]
We are very excited about the addition of the cetrorelix DMF to our growing generic peptide portfolio. Our multi-kg scale cGMP manufacturing facility for cetrorelix will provide more opportunities for IVF treatments in the medical communities and pathways to new treatments for hormone-sensitive breast and prostate cancers.
Nakagawa, Mayumi. U.S. Patent Application 15/552,285, filed February 15, 2018.
“The PepCan peptide mixture will contain four HPV 16 E6 peptides: E6 1-45 (Ac-MHQKRTAMFQDPQER PRKLPQLCTELQTTIHDIILECVYCKQQLL-NH2..), E6 46-80 (Ac-RREVYDFAFRDLCIV YRDGN PYA VCDKCLKFYSKI-NH2..), E6 81-115 (Ac-SEYRHYCYSLYGTTLEQQYNK PLCDLLIRCINCQK-NH2..), and E6 116-158 (Ac-PLCPEEKQRHLDKKQRFHNIRGRWT GRCMSCCRSSRTRRETQL-NH2..) (U.S. Pat. No. 8,652,482). Commercially produced cGMP-grade peptides (CPC Scientific, San Jose, Calif.) will be examined..”
Joshi, Bishnu P., et al. Endoscopy 48.02 (2016): A1-A13
"ASY*-FITC was synthesized using good manufacturing practice (GMP) methods (CPC Scientific, Inc.). Stability was evaluated on high-performance liquid chromatography (HPLC) and mass spectrometry.”
Nakagawa, Mayumi. U.S. Patent Application No. 14/566,604, filed August 13, 2015.
“To define the minimal and optimal amino acids sequences of the CD8 T-cell epitope, 8-mer, 10-mer, 11-mer, and homologous peptides were synthesized (CPC Scientific, Inc, San Jose, Calif.)… Their safety for human use is ensured, in accordance with FDA requirements…(each peptide to be assessed individually […]
Saenz, Rebecca, et al. J Translational Medicine 12 (2014): 211.
“…Hp91 (DPNAPKRPPSAFFLFCSE), Hp121 (SIGDVAKKLGEMWNNTAA), scrambled Hp91 (ASLAPPFPNCFDPKSREF), and OVA-I (SIINFEKL) were all synthesized at GMP facilities by [..] CPC Scientific (San Jose, CA, USA). Peptides were synthesized with an N-terminal biotin, acetyl, or fluorescent tag (Cp488) as indicated in the figure legends. Peptides were routinely synthesized with greater than 95% purity.”
Wang, Xuelian, et al. Vaccine 31.49 (2013): 5806-5813.
“…four current good manufacturing practice-grade HPV16 E6 peptides [E6 1-45, E6 46-80, E6 81-115, and E6 116-158 (referred to as “peptides” hereafter); 10 μg/ml/peptide; made by CPC Scientific, Sunnyvale, CA…”
Sturm, Matthew B., et al. Science Translational Medicine 5.184 (2013): 184ra61-184ra61.
“For the clinical and pharmacology/toxicology studies, ASY*-FITC was GMP synthesized (CPC Scientific). Stability was evaluated by appearance (color), HPLC, and mass spectrometry for 3 years. The pharmacology/toxicology study in rats was conducted in accordance with FDA CFR title 21, part 58…”






