Commonly Used Oligonucleotide Abbreviations
- DNA = A, C, G, T, 5MeC [for 5-Me-dC]
- RNA = rA, rG, rC, rU
- 2′ O-Methyl (2’-OMe) = mA, mG, mC, mU, m5MeC [for 2′-OMe, 5-Me C]
- 2ʹ-O-Methoxyethyl (2’-MOE) = moeA, moeG, moeC, moeU, moe5MeC, moe5MeU [for 2ʹ-moe 5-methyl cytidine and uridine]
- Locked (2’-LNA) = lnA, lnG, lnC, lnU, ln5MeC, ln5MeU [for locked RNA 5-methyl cytidine and uridine]
- 2′-F = fA, fG, fC, fU
- Backbone bases: Phosphodiester bases = (PO), Phosphorothioate = (PS), Phosphoroamidate = (PN)
Standard Testing
- Appearance Visual White to off-white powder
- Purity HPLC NLT 85% or NLT 90%; as requested
- Identity, MW ESI-MS +/- 0.05% [3Da for theoretical MW of 6,000]
| A Adenine | C Cytosine | T/U Uracil / Thymine | G Guanine |
|
|---|---|---|---|---|
| DNA | A | C | T | G |
| RNA | rA | rC | U | rG |
| 2'-OMe | mA | mC | mT/U | mG |
| 2'-MOE | moeA | moeC | moeT/U | moeG |
| 2'-cEt | cEtA | cEtC | cEtT/U | cEtG |
| Locked (2'-LNA) | lnA (or +A) | lnC (or +C) | lnT/U (or +T/U) | lnG (or +G) |
| 2'-F | fA | fC | fT/U | fG |
| PO backbone = Phosphodiester (Standard), no indication necessary. | A | C | T/U | G |
| PS backbone = phosphorothioate, indicated with an * in between bases. | *A | *C | *T/U | *G |
| PN backbone = phosphoramidate, indicated with an ^ in between bases. | ^A | ^C | ^T/U | ^G |
Canonical Amino Acids
| A Ala ALANINE | C Cys CYSTEINE | D Asp ASPARTIC ACID | E Glu GLUTAMIC ACID | F Phe PHENYLALANINE | G Gly GLYCINE | H His HISTIDINE | I Ile ISOLEUCINE | K Lys LYSINE | L Leu LEUCINE |
| M Met METHIONINE | N Asn ASPARAGINE | P Pro PROLINE | Q Gln GLUTAMINE | R Arg ARGININE | S Ser SERINE | T Thr THREONINE | V Val VALINE | W Trp TRYPTOPHAN | Y Tyr TYROSINE |
Chemical Structures of Common Amino Acids

Chemical Structures of Unnatural Amino Acids
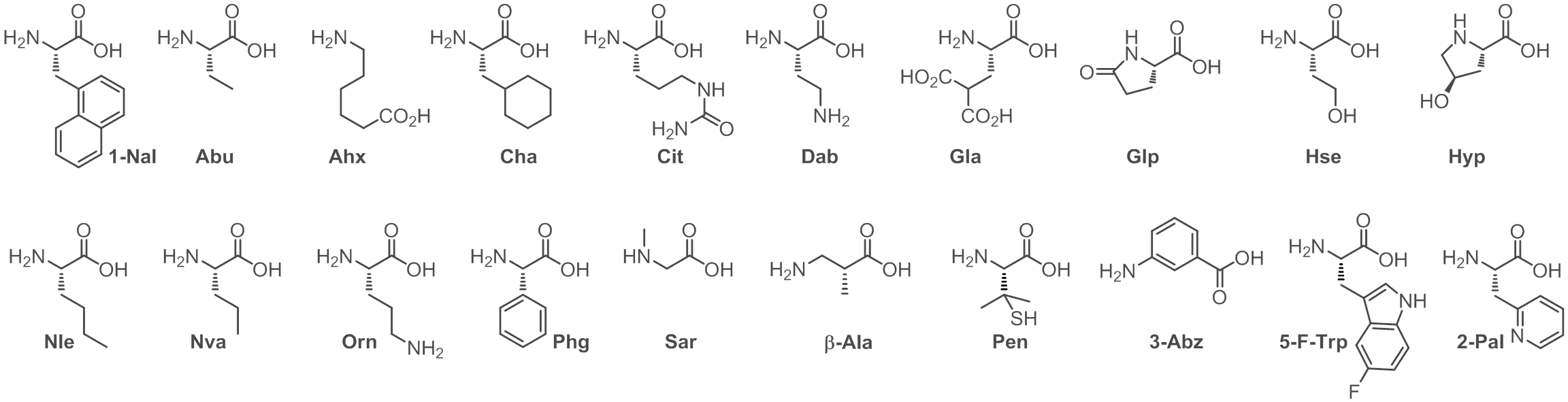
Table of Common Unnatural Amino Acids
| 1-Nal β-(1-Naphthyl)-L-alanine | Dab L-α,γ-Diaminobutyric acid | Nle Norleucine | Phg L-Phenylglycine |
| Abu 2-Aminobutyric Acid | Gla Carboxyglutamic Acid | Nva Norvaline | Sar Sarcosine (N-Methylglycine) |
| Ahx 6-Aminohexanoic acid | Glp Pyroglutamic Acid | Orn Ornithine | β-Ala β-Alanine |
| Cha Cyclohexylalanine | Hse Homoserine | Pal 2-Pyridylalanine | Cit L-Citrulline |
| Hyp trans-4-Hydroxy-L-proline | Pen L-Penicillamine | Tle tert-butyl-Leucine | Fpa 3-Fluoro-Phenylalanine-L-Alanine |
CPC Scientific can indeed aliquot any order to your specifications. By aliquotting your order into smaller quantities, this service will allow you to store and access your peptide with ease and also gives you the ability to standardize your protocols. Perhaps most important, the peptide will enjoy greater stability in the long run by preventing contamination from the frequent opening of the container. Repeated thawing/freezing and opening/closing of the container will invariably introduce moisture into the system. This could lead to bacterial degradation, peptide oxidation, and aggregation, lowering the purity of your peptide and causing loss of continuity in future experiments. Weights for aliquots 1 mg and over will be determined by our balance scale. All aliquots less than 1 mg will be aliquoted volumetrically for more precision than our balance offers.
CPC performs a very specialized pooling service for its customers. We can customize this to suit any customer’s needs, but we have a general protocol that is very effective at demonstrating the contents of a pool. First, subpools of 20 peptides are created. MS is run on these pools to display each peak for each peptide contained in the subpool. In most cases, the peptides are of a different enough mass to clearly point out each peak. However, if the masses of the two peptides are very similar, it may be difficult or impossible to discern. After all of the subpools have been created, they are then combined into a master pool. Finally, MS is performed on the master pool with the notion of identifying one peak from each individual subpool. In doing so, this demonstrates that each subpool and subsequently each peptide are present in the master pool.
CPC can produce all commercially available modifications with the exception of radiolabeling. CPC is happy to provide you with answers to any of your technical questions with regard to modification. We can make virtually any synthetic modification to a peptide and are not limited to following:
- FRET Substrates Design
- Isotopic Labeling
- Peptide Stapling
- Macrocyclization
- Clickable Peptides
- Dye Labeling
- PEGylation
- Chelate Conjugation
- Glycosylation
- Phosphorylation
- Sulfation and Sulfonation
- Unnatural Amino Acids
- Lipidation
- Hydrophobic Sequences
- Peptoids
- Rhodamine 110 Labeling
- Cell-Penetrating Peptides
- Long Sequences
- Native Chemical Ligation
- Epitope Mapping
- Peptide Libraries
- Neoantigen-based Libraries
- Protein Conjugation
- Selenocysteine
- Depsipeptides
- Peptidomimetics
- Multiple Antigenic Peptides (MAPs)
- Maleimide Group Installation
Catalog peptides are commercially available, published sequences. Commonly used catalog peptides are frequently produced in bulk at purities exceeding 95%, and they are generally kept in-stock. Producing these peptides in larger quantities allows companies to sell them at lower prices due to the bulk process involved. Custom peptides, on the other hand, are not public domain. Certain changes or modifications to catalog peptides must be ordered as a custom order for this reason. When ordering a custom peptide, the 2 most determinate factors are purity needed and sequence length. The time length involved in producing a custom peptide at CPC is generally 2-3 weeks.
Every peptide (including crude) is shipped with HPLC and MS. A bonus that CPC provides with its QC package is a recommended solubility test on each peptide. This solubility test utilizes solvents that are applicable for your experiment (water is the first choice), and can be customized according to the customer’s specifications. The test will save you time and effort in trying to determine the solubility and will also save peptide because you won’t need to use a small sample for testing if you do not have the ability to lyophilize the peptide.
All peptides are shipped directly to the end-user from our Rocklin, CA office by FedEx overnight service. On the day of the shipment, an e-mail is sent to the end-user confirming the shipment and providing a FedEx number in order to track the package if needed. Each peptide is shipped at ambient temperature and accompanied with desiccant to absorb excess moisture. Various tests have revealed shipping under these conditions has no impact on the viability and shelf-life of the peptides. However, upon receipt of the product by the end user, it should be stored immediately at -20 C or lower.
CPC Scientific has produced a peptide of 130 amino acids in length, but in general, peptides of more than 30 amino acids can be problematic. Solubility is one of the most important factors to consider when designing a peptide as it will affect the purity attained since HPLC is performed in solution. The more hydrophobic residues in the sequence, the less soluble the peptide is. Certain sequences can also aggregate which will lead to difficulties in production. CPC has over 100,000 peptides worth of peptide design experience at your disposal, and we are happy to assist any questions.
The crucial link in peptide synthesis is the peptides bond formed by condensation of the alpha-amino group of one amino acid and carboxyl group of another amino acid. Amino acids contain a central carbon atom surrounded by hydrogen, an amino group, a carboxyl group, and a side chain group, designated as R. The R group is what defines each individual amino acid and determines the physical and chemical properties of the amino acid. Certain R groups can interfere with the formation of the peptide bond, and, therefore, it is necessary to orthogonally protect the functional side chains of certain amino acids. Different protecting groups are available depending on the type of chemistry used and the application. Synthetic peptides differ from natural protein synthesis on ribosomes in that the synthesis is conducted from the C to N terminuses. In solid-phase peptide synthesis, the first amino acid is covalently linked to a solid support with the alpha-amino group protected by either a Fmoc or t-Boc moiety. Treatment with a deprotection agent (piperidine for Fmoc, TFA for Boc) frees the alpha-amino group in preparation for coupling the next amino acid in the sequence. The next amino acid is activated by one of several reagents and a peptide bond is formed. This freshly coupled amino acid is protected on its alpha-amino group as well, and in this way prevents double addition of the amino acid into the sequence. This stepwise addition continues until the desired peptide length is obtained. After the last amino acid has been added, it is necessary to perform one additional deprotection step to remove the last moiety on the N terminal amino acid. Once the synthesis has been completed, the peptide must be removed from the solid support. This is accomplished by adding an appropriate acid (TFA for Fmoc, HF for Boc) as well as scavengers to collect protected functional groups.
When producing peptide-antibodies, it is important to use a carrier protein to elicit a better response from the animal. Commonly used carrier proteins are BSA and KLH, with KLH preferred for its higher immunogenicity. Peptides coupled with BSA will generate antibodies against both the peptide and the BSA, which could result in incorrect ELISA readings. For coupling carrier proteins to peptides, we recommend incorporating an N terminus cysteine for this reaction due to the high efficiency and proven effectiveness. For coupling through cysteine, we use MBS (maleimidobenzoic acid-N-hydroxysuccinimide ester) as a crosslinker. The initial step is to activate the protein with MBS, and then remove the excess crosslinker by SEC. This activated carrier protein is then coupled to the cysteine in the peptide through a disulfide bond with an efficiency of >95%. The only impurity in this reaction is salt formation, which we remove through gel filtration. As an alternative to coupling through cysteine, we can additionally couple the carrier protein of choice through an amino link. The coupling efficiency of this reaction is generally around 80%. For carrying out this reaction, glutaraldehyde is used to crosslink the amino groups in both peptide and carrier protein. The most undesirable part of this reaction is the fact that protein-protein and peptide-peptide crosslinking can occur, and for this reason, the most important step is to insure that the proper concentration of crosslinker.
The reported purity of a peptide is a percentage of the target sequence as determined by HPLC. The impurities found in a peptide will include deletion sequences, truncated sequences, and incompletely deprotected sequences. Peptide content can be determined in a few different ways depending on the sequence and is a measurement of the native peptide present versus the non-peptide components.
All peptides are delivered by gross weight unless otherwise specified. Gross peptide weight is the weight of the peptide and peptide impurities as well as components such as salts, counter ions, residual solvents, and water. Net peptide, on the other hand, is a calculation of merely the peptide present. In certain experiments, this can be critical information as the peptide content can vary widely depending primarily on the sequence.
Immunological grade and > 70% purity
- Antigen for antibody production
- ELISA standard for measuring titers of antibodies in antisera
- Competitive elution chromatography
- Peptide array production
80% purity
- Non-quantitative enzyme-substrate studies
- Phosphorylation assays
- Non-quantitative peptide blocking studies
- Coupling to resins for affinity purification
- Coating of tissue culture plates for cell attachment
- Protein electrophoresis applications
95% purity
- Standards for quantitative ELISA and RIA protocols
- Quantitative receptor-ligand interaction studies
- In vitro bioassays
- In vivo studies
- Quantitative enzyme studies
- NMR studies
- Mass spectrometry
- Quantitative assays
98% purity
- SAR Studies
- Clinical trials
- API (Active Pharmaceutical Ingredients)
- Commercial products
- X-Ray crystallography studies
Because peptides are hygroscopic, it is important to allow the peptide to warm to Romm temperature prior to opening. Warming in a desiccator or dry box is the best way to avoid water condensation forming on the peptide. Weigh out the prodcut quickly in a clean environment only the needed quantity to be used to minimize contamination and reseal containers tightly. Store all unused peptide at -20 C or less in a sealed container with desiccant. Storage under these conditions will reduce and prevent bacterial degradation, secondary structure formation, and oxidation for up to several years. Sequences containing cysteine, methionine, tryptophan, asparagine, glutamine, and N-terminal glutamic acid will have a shorter shelf life than other peptides. When solubilizing the peptide, please use the recommended solvents outlined in the solubility test provided by CPC if your experimental conditions will allow it. Sonication is often helpful to dissolve the peptide. In general, sterilized water is the preferred solvent, but acidic peptides will dissolve in a basic buffer and basic peptides in an acidic buffer as determined by the amino acid sequence of the peptide. Peptide sequences containing Cys, Met, and Trp require oxygen-free solvents as they are especially prone to oxidation. Peptides in solution are much less stable than lyophilized form and will remain viable for a much shorter period of time. As such, it is not recommended to store peptides under these conditions, but if absolutely unavoidable, the peptides in solution should be kept at -20 C using sterile buffers at pH 5-6 to prolong shelf life. The best way to avoid running into this issue is to have CPC aliquot the peptide.
