Enabling Sustainable Large-Scale SPPS Through Process Innovations
Delivery of polypeptides in multi-kilogram quantities within tight timescales is extremely challenging, especially when coupled with a desire to minimize environmental and economic impact. This case study explores how CPC Scientific’s advanced process improvements during synthesis, cleavage, and final purification of solid-phase peptide synthesis (SPPS) have enabled multi-kilogram delivery of a pharmaceutically-relevant decapeptide within a short timescale. These innovative modifications reduced environmental footprint and delivered substantial cost savings for the client, ultimately enhancing market competitiveness of the product.
Delivery of polypeptides in multi-kilogram quantities within tight timescales is extremely challenging, especially when coupled with a desire to minimize environmental and economic impact. This case study explores how CPC Scientific’s advanced process improvements during synthesis, cleavage, and final purification of solid-phase peptide synthesis (SPPS) have enabled multi-kilogram delivery of a pharmaceutically-relevant decapeptide within a short timescale. These innovative modifications reduced environmental footprint and delivered substantial cost savings for the client, ultimately enhancing market competitiveness of the product.

Download White Paper Form
Partner with CPC Scientific to scale your projects with greener, more efficient manufacturing
With 25 years of CDMO experience in the pharmaceutical industry, CPC Scientific is committed to transforming advanced science into commercial-ready therapeutic solutions for patients worldwide.
Ready to explore innovative manufacturing solutions that drive sustainability without compromising on quality? Reach out below, or connect with our experts at sales@cpcscientific.com today.
Partner with CPC Scientific to scale your projects with greener, more efficient manufacturing
With 25 years of CDMO experience in the pharmaceutical industry, CPC Scientific is committed to transforming advanced science into commercial-ready therapeutic solutions for patients worldwide.
Ready to explore innovative manufacturing solutions that drive sustainability without compromising quality? Reach out below, or connect with our experts at sales@cpcscientific.com today.
READ OUR LATEST WHITE PAPERS
Recent peptide- and oligonucleotide-based articles designed to increase your understanding about various subject matters.
Recent peptide- and oligonucleotide-based articles designed to increase your understanding about various subject matters.
Delivery of polypeptides in multi-kilogram quantities within commercially competitive timelines is extremely challenging, especially when coupled with a desire to minimize environmental and economic impact. This case study explores how CPC Scientific’s advanced process improvements and efficiencies within solid-phase peptide synthesis (SPPS) have enabled multi-kilogram delivery of a pharmaceutically-relevant decapeptide within a challenging timescale, driving sustainable and cost-saving production for the client.
Our team has developed an innovative DMF recycling strategy that substantially reduces solvent consumption during solid-phase peptide synthesis. Minimizing use of DMF, a major environmental and cost contributor in peptide manufacturing, has improved process sustainability and cost efficiency. This method neatly demonstrates how targeted green chemistry practices can be successfully integrated into large-scale SPPS, supporting more environmentally responsible and economically viable peptide production.
In Part 1 of our Minimal Protection Group Strategies for SPPS, we discussed methods for eliminating sidechain protection on hydroxy-bearing amino acids such as serine, threonine, tyrosine, and hydroxyproline. By omitting t-butyl protection, we enhanced atom economy and avoided the use of hazardous solvents typically required to remove these protection groups. In Part 2, we present a new case study, expanding our approach to include the unprotected side chains of histidine, tryptophan, and arginine. We demonstrate the synthesis of a Goserelin peptide API impurity, showcasing how a convergent peptide fragment strategy can be used to eliminate the need for TFA and diethyl ether, eliminate side chain protection of Arginine, Histidine, and Tryptophan.
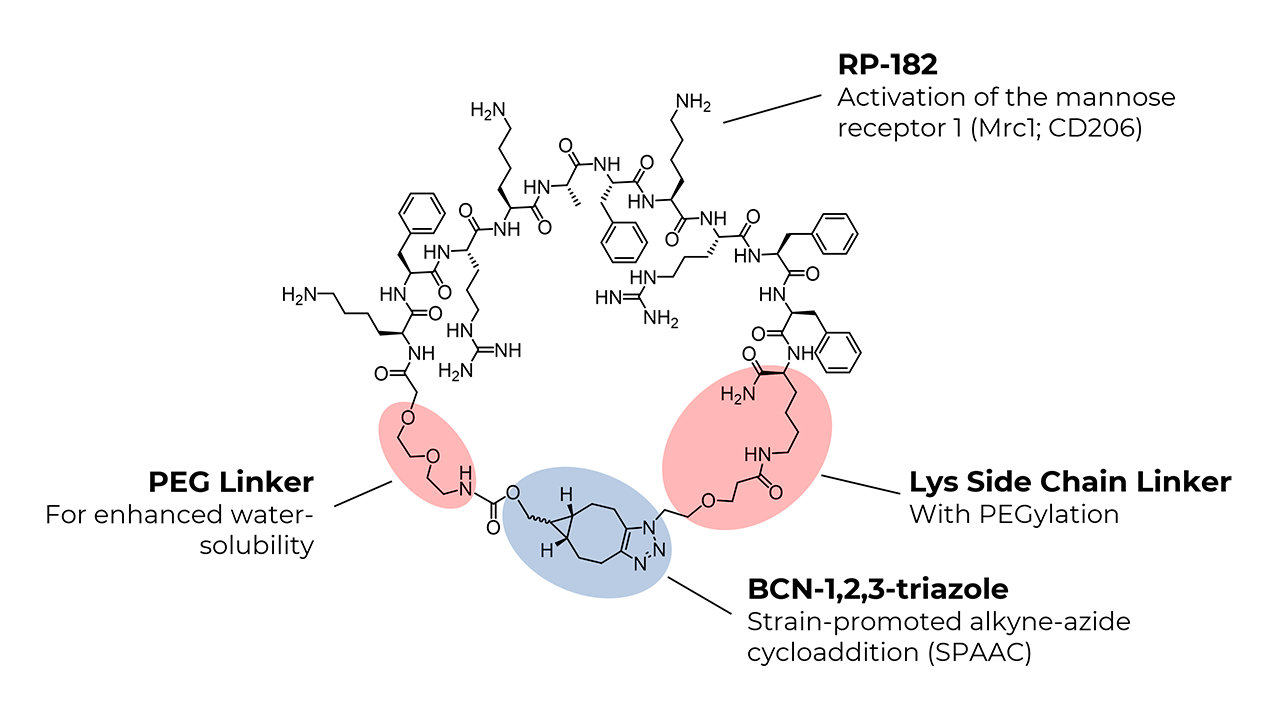
The synthesis of the linear RP-182 analog, bicyclo[6.1.0]non-4-yn-9-ylmethyloxycarbonyl-PEG2-Lys-Phe-Arg-Lys-Ala-Phe-Lys-Arg-Phe-Phe-Lys(azido-PEG)-NH2, was achieved using standard solid-phase peptide synthesis (SPPS) protocols. After cleaving the linear peptide from the resin, macrocyclization was performed in the liquid phase through a strain-promoted click reaction. BCN introduces extra ring strain due to its fused cyclopropane structure. The combined effect of ring strain, the selection of BCN, and copper catalysis significantly increases the macrocyclization efficiency of longer peptides like RP-182.
Stapled peptides have emerged as a powerful tool in drug discovery and therapeutic development due to their ability to overcome the limitations associated with traditional peptide drugs, such as poor stability and low cell permeability. By introducing staples into the peptide backbone, researchers can stabilize peptide conformations and enhance their interactions with target proteins, resulting in improved efficacy and specificity. This approach not only addresses the challenges of peptide drug design but also opens new avenues for targeting challenging biomolecular interactions that are difficult to modulate with small molecules or antibodies. The development of stapled peptides has led to significant advancements in targeting protein-protein interactions, addressing previously intractable diseases, and enhancing the precision of therapeutic interventions.
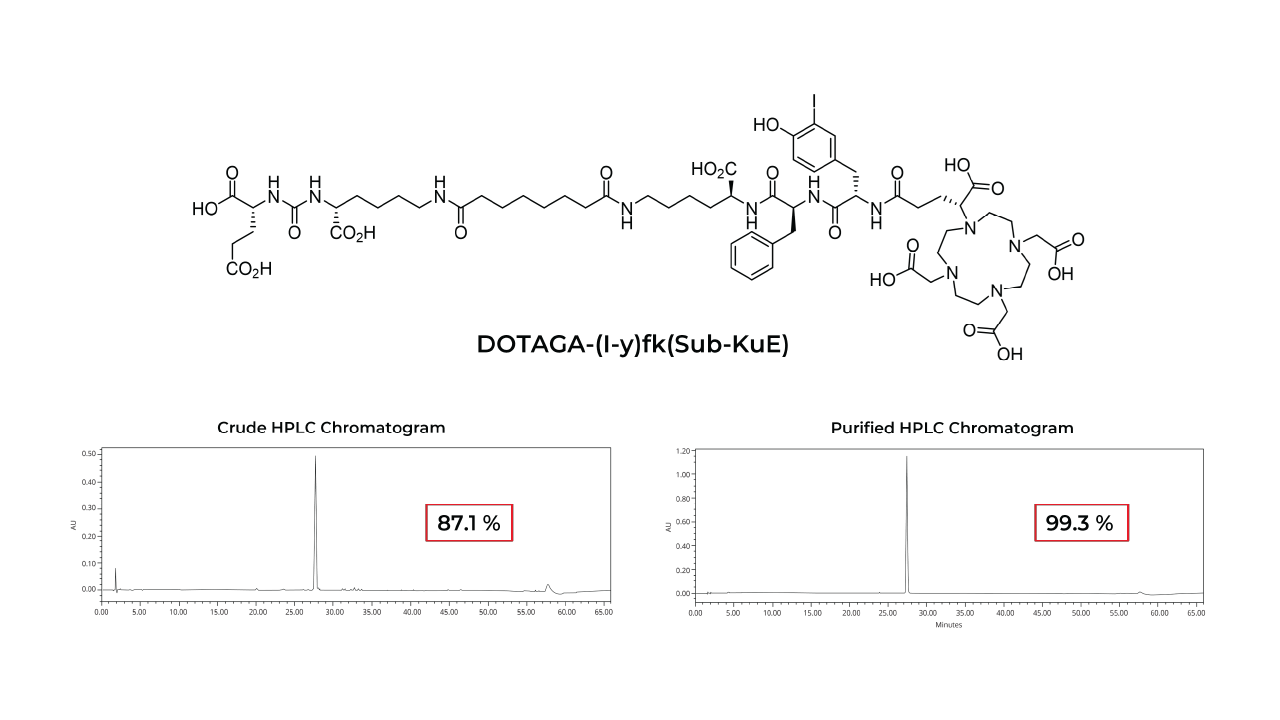
Peptide receptor radionuclide therapy (PRRT) is a targeted therapeutic approach that utilizes peptides to deliver cytotoxic radiation to specific receptors overexpressed on cancer cells. Peptides offer several advantages as therapeutic vectors in PRRT due to their small size, favorable pharmacokinetics, high binding affinity, low immunogenicity and toxicity, and minimal off-target effects. Tumor-targeting peptides conjugated to radionuclide chelates represent a promising class of cancer therapeutics.
Delivery of polypeptides in multi-kilogram quantities within commercially competitive timelines is extremely challenging, especially when coupled with a desire to minimize environmental and economic impact. This case study explores how CPC Scientific’s advanced process improvements and efficiencies within solid-phase peptide synthesis (SPPS) have enabled multi-kilogram delivery of a pharmaceutically-relevant decapeptide within a challenging timescale, driving sustainable and cost-saving production for the client.
Our team has developed an innovative DMF recycling strategy that substantially reduces solvent consumption during solid-phase peptide synthesis. Minimizing use of DMF, a major environmental and cost contributor in peptide manufacturing, has improved process sustainability and cost efficiency. This method neatly demonstrates how targeted green chemistry practices can be successfully integrated into large-scale SPPS, supporting more environmentally responsible and economically viable peptide production.
In Part 1 of our Minimal Protection Group Strategies for SPPS, we discussed methods for eliminating sidechain protection on hydroxy-bearing amino acids such as serine, threonine, tyrosine, and hydroxyproline. By omitting t-butyl protection, we enhanced atom economy and avoided the use of hazardous solvents typically required to remove these protection groups. In Part 2, we present a new case study, expanding our approach to include the unprotected side chains of histidine, tryptophan, and arginine. We demonstrate the synthesis of a Goserelin peptide API impurity, showcasing how a convergent peptide fragment strategy can be used to eliminate the need for TFA and diethyl ether, eliminate side chain protection of Arginine, Histidine, and Tryptophan.
The synthesis of the linear RP-182 analog, bicyclo[6.1.0]non-4-yn-9-ylmethyloxycarbonyl-PEG2-Lys-Phe-Arg-Lys-Ala-Phe-Lys-Arg-Phe-Phe-Lys(azido-PEG)-NH2, was achieved using standard solid-phase peptide synthesis (SPPS) protocols. After cleaving the linear peptide from the resin, macrocyclization was performed in the liquid phase through a strain-promoted click reaction. BCN introduces extra ring strain due to its fused cyclopropane structure. The combined effect of ring strain, the selection of BCN, and copper catalysis significantly increases the macrocyclization efficiency of longer peptides like RP-182.
Stapled peptides have emerged as a powerful tool in drug discovery and therapeutic development due to their ability to overcome the limitations associated with traditional peptide drugs, such as poor stability and low cell permeability. By introducing staples into the peptide backbone, researchers can stabilize peptide conformations and enhance their interactions with target proteins, resulting in improved efficacy and specificity. This approach not only addresses the challenges of peptide drug design but also opens new avenues for targeting challenging biomolecular interactions that are difficult to modulate with small molecules or antibodies. The development of stapled peptides has led to significant advancements in targeting protein-protein interactions, addressing previously intractable diseases, and enhancing the precision of therapeutic interventions.
Peptide receptor radionuclide therapy (PRRT) is a targeted therapeutic approach that utilizes peptides to deliver cytotoxic radiation to specific receptors overexpressed on cancer cells. Peptides offer several advantages as therapeutic vectors in PRRT due to their small size, favorable pharmacokinetics, high binding affinity, low immunogenicity and toxicity, and minimal off-target effects. Tumor-targeting peptides conjugated to radionuclide chelates represent a promising class of cancer therapeutics.